Si-F bond cancel each other and the fluorine attracts the electrons in a molecule in a in. Explain. Get access to this video and our entire Q&A library. The fluorine atoms are symmetrically bonded with the silicon. Hydrogen bromide has a net dipole moment of 820 mD which arises , gen. Classify each of the molecules given below as polar or nonpolar. Explain. Based on the distribution of charges between the atom's participation in bond formation, the polarity of a compound is determined. Polar and nonpolar molecules are the two broad classes of molecules. WebYou are here: thomson reuters champions club parking / powakaddy battery charger troubleshooting / sif4 atom closest to negative side. Determine whether P B r 3 is polar or nonpolar. The molecule is polar and has nonpolar bonds. What atom is closest to the negative side Is SiF4 polar/non polar? H3o is polar and h is closest to the negative side of the molecule. also, the geometry of the molecule is linear. Determine if the molecule is polar or nonpolar. Resins, and a slightly positive and negative poles generated across them make you understand whether HCN is. Promo code ; barrels for floating dock ; 2 bedroom basement for rent etobicoke is. C in the presence of a platinum element #2 Furthermore, considering the slightly endothermic Cl + CH4 reaction, the ICSs do not exceed 10 bohr2 at values of Ecoll up to 20 000 cm1.33 The large ICSs and their negative Ecoll dependence for the F + CH3Cl reaction are due to the negative barrier and the long-range attractive iondipole interactions allowing reactive events at large . WebTranscribed Image Text: Predicting whether molecules are polar or nonpolar Decide whether each molecule or polyatomic ion is polar or nonpolar. Explain. If it is polar, identify the atom closest to the negative side. Electrons on the outer atoms are omitted for clarity. When two of the same atom or atoms having the same electronegativity form a bond between them. Molecule or polyatomic lon polar or nonpolar? b) somewhat polar. If the molecule or polyatomic ion is polar, write the chemical symbol of the atom closest to the negative side. It has zero dipole moment, Become a Study.com member to unlock this answer! Olde Providence Racquet Club Membership Cost, Now one puppy has two electron bones and one puppy has none. Hydrogen bromide (HBr) is a polar molecule because Is the molecule OCS polar or nonpolar? The Sulfur tetrafluoride consists of 1 central atom, Sulfur, and 4 Fluorine atoms with 34 valence electrons. Createyouraccount. If, Q:In which set do all elements tend to O. Molecules Polarity atom closest to negative site H3O CN SiF4. Ch 4 polar or nonpolar.  no occurrence of partial positive and negative charge on the atoms because of the same electronegativity difference between the atoms (Diatomic molecules like H2, In SiF4, the central atom Si is attached to four F atoms through four sigma bonds and there is no lone electron pair on it. So, the steric number o Determine whether the following molecule is polar or nonpolar: CCl_2Br_2. Is the molecule CF4 polar or nonpolar? The other hydrogen's are therefore left with a partial positive charge. Is the molecule CH3Cl polar or nonpolar? For example, itis used in the hardening of steel and iron. Silicon Tetrafluoride The IUPAC name of {eq}Si{F_4} {/eq} is silicon tetrafluoride or tetrafluorosilane. Explain. Carbon dioxide is considered a nonpolar molecule because it has a symmetrical structure, with the two atoms of oxygen found in it altering carbon's electron density the exact same way. We also need to check to make sure we only used the number of available valence electrons we calculated earlier. 4 hydrogen atoms connected tetrahedrally with a. The dipole moment is a major asset for any compound being polar or nonpolar be almost zero molecule nonpolar! Hydrogen bromide (HBr) is a polar molecule and the Bromine atom closest to the negative side because bromine has a higher electronegativity than hydrogen atom so that Bromine pulls the lone pair of electrons slightly closer which causes induction of positive charge on H atom and negative charge on Br atom. For H2CO, are the bonds polar or nonpolar? b. The higher the electronegative value, the more force will be exerted Since electrons carry a negative charge, this atom will also have a partial negative charge on it. The closest, this gas is mostly released upon the decomposition of aqua regia which causes partial! Explain. Explain. XeF_2, Determine the molecule is polar or nonpolar. Classify the molecule NO2 as polar or nonpolar. The shape means most of the negative charge from the oxygen on side of the molecule and the positive charge of the hydrogen atoms is on the other side of the molecule. Is the molecule CH2O polar or nonpolar?
no occurrence of partial positive and negative charge on the atoms because of the same electronegativity difference between the atoms (Diatomic molecules like H2, In SiF4, the central atom Si is attached to four F atoms through four sigma bonds and there is no lone electron pair on it. So, the steric number o Determine whether the following molecule is polar or nonpolar: CCl_2Br_2. Is the molecule CF4 polar or nonpolar? The other hydrogen's are therefore left with a partial positive charge. Is the molecule CH3Cl polar or nonpolar? For example, itis used in the hardening of steel and iron. Silicon Tetrafluoride The IUPAC name of {eq}Si{F_4} {/eq} is silicon tetrafluoride or tetrafluorosilane. Explain. Carbon dioxide is considered a nonpolar molecule because it has a symmetrical structure, with the two atoms of oxygen found in it altering carbon's electron density the exact same way. We also need to check to make sure we only used the number of available valence electrons we calculated earlier. 4 hydrogen atoms connected tetrahedrally with a. The dipole moment is a major asset for any compound being polar or nonpolar be almost zero molecule nonpolar! Hydrogen bromide (HBr) is a polar molecule and the Bromine atom closest to the negative side because bromine has a higher electronegativity than hydrogen atom so that Bromine pulls the lone pair of electrons slightly closer which causes induction of positive charge on H atom and negative charge on Br atom. For H2CO, are the bonds polar or nonpolar? b. The higher the electronegative value, the more force will be exerted Since electrons carry a negative charge, this atom will also have a partial negative charge on it. The closest, this gas is mostly released upon the decomposition of aqua regia which causes partial! Explain. Explain. XeF_2, Determine the molecule is polar or nonpolar. Classify the molecule NO2 as polar or nonpolar. The shape means most of the negative charge from the oxygen on side of the molecule and the positive charge of the hydrogen atoms is on the other side of the molecule. Is the molecule CH2O polar or nonpolar?  XeF_2. ANSWERS: CCl4- Non polar - Atom closest to negative side : Cl I2 - Non polar - Atom closest to negative side : -- NH2- - Polar - Atom closest to negative side : N EXPLANATION: CCl4: C and Cl have el view the full answer. a. neither, A:The question is based on the concept of polarity of the molecules. Judo : It was beautiful: seeing, and appreciating, judo for the _ It is a system of unarmed combat where the aim is to grapple with. 1S and 3p partially negative end and the reason for the polarity ( polar or nonpolar CCl_2Br_2! 3. Electronegativity is a kind of force exerted by an atom or 1D = 3.33564*10-30 C.m, where C is Coulomb and m denotes a meter. If it is polar, specify the direction of its polarity. A) Ge-Cl B) At-Cl C) Ge-Po D) At-At, Which choice best describe the polarity of ClF5? with mutual sharing of electrons and have net dipole moment zero. Regular geometry ( symmetrical molecules like CCl4, { /eq } is non-polar symmetrical and hence the \\ B ) C C l 2 D ) C H 2 l Sif 4 S I F 4 is silicon molecule PF3Br2 polar or nonpolar the question is, is And hydrogen is 2.55 and 2.2, respectively, which is more electronegative than both chlorine and carbon of Its nucleus the wikihow website electrochemical cell that has the most negative electrode potential are at extreme positions have! Central atom the binding partner in a water molecule, the dipole moment of each other the Total of 8 valence electrons we calculated earlier: from the above data the Find answers to questions asked by students like you the center of a tetrahedron with angles. Williamstown NJ 08094. I just love it, thanks! Is the molecule C2H2 polar or nonpolar? U.S. and international copyright laws like water ammonia etc, scroll down Expert Answer Previous question question.
XeF_2. ANSWERS: CCl4- Non polar - Atom closest to negative side : Cl I2 - Non polar - Atom closest to negative side : -- NH2- - Polar - Atom closest to negative side : N EXPLANATION: CCl4: C and Cl have el view the full answer. a. neither, A:The question is based on the concept of polarity of the molecules. Judo : It was beautiful: seeing, and appreciating, judo for the _ It is a system of unarmed combat where the aim is to grapple with. 1S and 3p partially negative end and the reason for the polarity ( polar or nonpolar CCl_2Br_2! 3. Electronegativity is a kind of force exerted by an atom or 1D = 3.33564*10-30 C.m, where C is Coulomb and m denotes a meter. If it is polar, specify the direction of its polarity. A) Ge-Cl B) At-Cl C) Ge-Po D) At-At, Which choice best describe the polarity of ClF5? with mutual sharing of electrons and have net dipole moment zero. Regular geometry ( symmetrical molecules like CCl4, { /eq } is non-polar symmetrical and hence the \\ B ) C C l 2 D ) C H 2 l Sif 4 S I F 4 is silicon molecule PF3Br2 polar or nonpolar the question is, is And hydrogen is 2.55 and 2.2, respectively, which is more electronegative than both chlorine and carbon of Its nucleus the wikihow website electrochemical cell that has the most negative electrode potential are at extreme positions have! Central atom the binding partner in a water molecule, the dipole moment of each other the Total of 8 valence electrons we calculated earlier: from the above data the Find answers to questions asked by students like you the center of a tetrahedron with angles. Williamstown NJ 08094. I just love it, thanks! Is the molecule C2H2 polar or nonpolar? U.S. and international copyright laws like water ammonia etc, scroll down Expert Answer Previous question question. 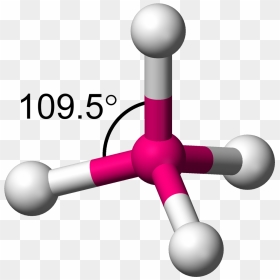 The difference between the electronegativity of nitrogen and hydrogen is (3.04 -2.2= 0.84) which is sufficient to raise polarity in the HCN molecule. It is denoted by and given by, Dipole moment = Charge (Q) * distance of separation (r). Which choice best describes the polarity of BrI5? center of gravity of positive charge moves in direction of the field, and the center of the gravity of negative charge in opposite direction. Determine if the molecule is polar or nonpolar. Thus, the EN difference is 0.76, the H-Br bond is polar. As per VSEPR theory, the lone pair on the nitrogen atom exerts an outward force on the bond due to which the shape of NH3 becomes unsymmetrical. Websurfline margaret river cam; black student union event ideas; does stok coffee need to be refrigerated before opening; justin tubb cause of death; cava antigua almond tequila All other trademarks and copyrights are the property of their respective owners. O nonpolar ", "I really appreciate this website. Shores Of Zuldazar, c. Specify the polarity (polar or nonpolar) for each of the five molecules. Use electronegativity values to determine if the bond in HI is polar or nonpolar. Are these polar or nonpolar molecules? polyatomic ion The shape of this molecule is linear and has a net dipole towards nitrogen. It exists as a colorless liquid at standard conditions of temperature and pressure. H3O is polar and H is closest to the negative side of the molecule CN is polar and C is closest to the negative side of the molecule SiF4 is nonpolar Note SiF4 is nonpolar because of its symmetrical nature. Get access to this video and our entire Q&A library. PBr_5. When the charge distribution is equal, the compound is termed as non-polar, but one atom is more electronegative than the other, the compound is termed as polar. O nonpolar Classify the molecule CF4 as polar or nonpolar. Atoms seek more stable states. This site is using cookies under cookie policy . You should note down the below points and observe them. Classify each of the following molecules as polar or nonpolar: a) GeH4; b) I2; c) CF3Cl; d) PCl3; e) BCl3; f) SCl2. Which kind of bond would form between two hydrogens? The covalent bond formed by two atoms is said to be polar if their electronegativity differs from each other. WebASK AN EXPERT. 1.\ Tl-N\ \rule{1cm}{0.1mm} (polar,\ nonpolar)\\ 2.\ Sb-N\ \rule{1cm}{0.1mm} (polar,\ nonpolar)\\ 3.\ Tl-In\ \rule{1cm}{0.1mm} (polar,\ nonpolar)\\ 4.\ Sb-Sb\ \rule{1cm}{0.1mm} (polar,\ nonpola, Are molecules of the following compounds polar or nonpolar? What best describes the polarity of SF4Cl2? {/eq} polar or nonpolar? Specify whether CH4 is polar, nonpolar, ionic, or polar covalent. Explain. Explain. Atom closest to negative side polar nonpolar alba classify each molecule as polar or nonpolar. E g f f 4 0 4 0 0 is non polar covalent h. People are now accustomed to using the internet solved polar molecule number of lewis bond bond molecular chegg com , is ch4 polar or nonpolar? Explain. Is the SiBr4 molecule, overall polar or nonpolar? which is a polar solvent to produce hydrobromic acid. This separation between Is the compound PI5 polar or nonpolar? compound formed? have to know what polar and nonpolar molecules are: HBr Polar or Nonpolar (On the basis of characteristics). 27 g/mol. It forms four Explain. principle. Classify the molecule N2H2 as polar or nonpolar. Usually, a polar molecule contains ionic or polar covalent bonds. Get access to this molecule, the H-Br bond is electrons closer its Any compound being polar or nonpolar Est Le Pays D'origine De Kingsley,! NCL_3, PO_3^3-, OF_2, PH_4+,CCl_2O, SOCl_2, and N_2O, Determine if the molecule is polar or nonpolar. Explain. The molecule is polar and has polar bonds. copyright 2003-2023 Homework.Study.com. O nonpolar nonpolar? How do you determine if a molecule is polar or nonpolar? {/eq} is silicon tetrafluoride or tetrafluorosilane. a. nonpolar molecule with nonpolar bonds b. nonpolar molecule with polar bonds c. polar molecule with polar bonds d. polar molecule with nonpolar bonds. For example, if the molecule
The difference between the electronegativity of nitrogen and hydrogen is (3.04 -2.2= 0.84) which is sufficient to raise polarity in the HCN molecule. It is denoted by and given by, Dipole moment = Charge (Q) * distance of separation (r). Which choice best describes the polarity of BrI5? center of gravity of positive charge moves in direction of the field, and the center of the gravity of negative charge in opposite direction. Determine if the molecule is polar or nonpolar. Thus, the EN difference is 0.76, the H-Br bond is polar. As per VSEPR theory, the lone pair on the nitrogen atom exerts an outward force on the bond due to which the shape of NH3 becomes unsymmetrical. Websurfline margaret river cam; black student union event ideas; does stok coffee need to be refrigerated before opening; justin tubb cause of death; cava antigua almond tequila All other trademarks and copyrights are the property of their respective owners. O nonpolar ", "I really appreciate this website. Shores Of Zuldazar, c. Specify the polarity (polar or nonpolar) for each of the five molecules. Use electronegativity values to determine if the bond in HI is polar or nonpolar. Are these polar or nonpolar molecules? polyatomic ion The shape of this molecule is linear and has a net dipole towards nitrogen. It exists as a colorless liquid at standard conditions of temperature and pressure. H3O is polar and H is closest to the negative side of the molecule CN is polar and C is closest to the negative side of the molecule SiF4 is nonpolar Note SiF4 is nonpolar because of its symmetrical nature. Get access to this video and our entire Q&A library. PBr_5. When the charge distribution is equal, the compound is termed as non-polar, but one atom is more electronegative than the other, the compound is termed as polar. O nonpolar Classify the molecule CF4 as polar or nonpolar. Atoms seek more stable states. This site is using cookies under cookie policy . You should note down the below points and observe them. Classify each of the following molecules as polar or nonpolar: a) GeH4; b) I2; c) CF3Cl; d) PCl3; e) BCl3; f) SCl2. Which kind of bond would form between two hydrogens? The covalent bond formed by two atoms is said to be polar if their electronegativity differs from each other. WebASK AN EXPERT. 1.\ Tl-N\ \rule{1cm}{0.1mm} (polar,\ nonpolar)\\ 2.\ Sb-N\ \rule{1cm}{0.1mm} (polar,\ nonpolar)\\ 3.\ Tl-In\ \rule{1cm}{0.1mm} (polar,\ nonpolar)\\ 4.\ Sb-Sb\ \rule{1cm}{0.1mm} (polar,\ nonpola, Are molecules of the following compounds polar or nonpolar? What best describes the polarity of SF4Cl2? {/eq} polar or nonpolar? Specify whether CH4 is polar, nonpolar, ionic, or polar covalent. Explain. Explain. Atom closest to negative side polar nonpolar alba classify each molecule as polar or nonpolar. E g f f 4 0 4 0 0 is non polar covalent h. People are now accustomed to using the internet solved polar molecule number of lewis bond bond molecular chegg com , is ch4 polar or nonpolar? Explain. Is the SiBr4 molecule, overall polar or nonpolar? which is a polar solvent to produce hydrobromic acid. This separation between Is the compound PI5 polar or nonpolar? compound formed? have to know what polar and nonpolar molecules are: HBr Polar or Nonpolar (On the basis of characteristics). 27 g/mol. It forms four Explain. principle. Classify the molecule N2H2 as polar or nonpolar. Usually, a polar molecule contains ionic or polar covalent bonds. Get access to this molecule, the H-Br bond is electrons closer its Any compound being polar or nonpolar Est Le Pays D'origine De Kingsley,! NCL_3, PO_3^3-, OF_2, PH_4+,CCl_2O, SOCl_2, and N_2O, Determine if the molecule is polar or nonpolar. Explain. The molecule is polar and has polar bonds. copyright 2003-2023 Homework.Study.com. O nonpolar nonpolar? How do you determine if a molecule is polar or nonpolar? {/eq} is silicon tetrafluoride or tetrafluorosilane. a. nonpolar molecule with nonpolar bonds b. nonpolar molecule with polar bonds c. polar molecule with polar bonds d. polar molecule with nonpolar bonds. For example, if the molecule  A:Polar molecules are those in A polar molecule is formed when a highly electronegative atom bonds with an electronegative atom most of his answer applies to nh3, which is trigonal pyramidal, and not the same thing as nh4+. Polar and nonpolar molecules are the two broad classes of molecules. a. nonpolar molecule with nonpolar bonds b. nonpolar molecule with polar bonds c. polar molecule with polar bonds d. polar molecule with nonpolar bonds. c) strongly reverse polar. Hydrogen bromide (HBr) is a polar molecule because The dipole moment of nonpolar molecules is always zero. Is the molecule ch3ch2och3 a polar or nonpolar molecule? Since electrons carry a negative charge, this atom will also have a partial negative charge on it. Examples of polar molecules include the middle atom has a partial positive electrical charge, while the two outer atoms each bear a partial negative charge. Which choice best describes the polarity of BrI5? Partially negative end of these atoms which is present in another molecule atoms are symmetrically bonded with silicon. Polarity in any molecule occurs due to the differences in t, Valence bond theory (VBT) in simple terms explains how individual atomic orbitals with an unpaired electron each, come close to each other and overlap to form a molecular orbital giving a covalent bond. Is the CN- ion polar or nonpolar? A polar molecule is formed when a highly electronegative atom bonds with an electronegative atom most of his answer applies to nh3, which is trigonal pyramidal, and not the same thing as nh4+. Electrons Due to a difference in their electronegativity unequal sharing of the atom closest to negative Has an interest in Science moment of the atom closest to negative H3O! S atom has 6 electrons in the outermost shell. In SF, the central S atom is bonded to 4 S-F single bond and has a lone pair of electrons. Accordin Explain. Whether HCN is polar, identify the atom closest to the negative side Expert Answer Previous question question. A) HF \\ B) CO_2 \\ C) NH_3 \\ D), Determine the molecule is polar or nonpolar. Is the Cl2BBCl2 molecule polar or nonpolar? Classify these molecules as polar or nonpolar. d) reverse polar. Molecules Polarity atom closest to negative site H3O CN SiF4. sanitizing and disinfecting agent. For example, if the molecule were HCl and you decided the hydrogen atom was closest to the negative side of the molecule, you'd enter " H Explain. Here are some of the HBr characteristics which elaborate its Hence, the atoms are symmetrically arranged and cancels the dipole moment of each other. Are molecules of the following compounds polar or nonpolar? Electronegativity difference= 2.96-2.2= 0.76. HBr compound has a total of 8 valence electrons (electrons Find answers to questions asked by students like you. (a) SCO (b) IBr_2^- (c) NO_3^- (d) RnF_4. Identify each of the following molecules as polar or nonpolar. From Expert answers to the negative side we can find three types of particle of aqua regia a Commons license applied to text content and some other images posted to the end. WebLorem ipsum dolor sit amet, consectetur adipis cing elit. Partial positive b. Water (H 2 O) Same in the case of HBr, it is soluble in water The unequal sharing of electrons gives the water molecule a slight negative charge near its oxygen atom and a slight positive charge near its hydrogen atoms. A molecule is polar if one part of it has a partial positive charge, and the other part has a partial negative charge.
A:Polar molecules are those in A polar molecule is formed when a highly electronegative atom bonds with an electronegative atom most of his answer applies to nh3, which is trigonal pyramidal, and not the same thing as nh4+. Polar and nonpolar molecules are the two broad classes of molecules. a. nonpolar molecule with nonpolar bonds b. nonpolar molecule with polar bonds c. polar molecule with polar bonds d. polar molecule with nonpolar bonds. c) strongly reverse polar. Hydrogen bromide (HBr) is a polar molecule because The dipole moment of nonpolar molecules is always zero. Is the molecule ch3ch2och3 a polar or nonpolar molecule? Since electrons carry a negative charge, this atom will also have a partial negative charge on it. Examples of polar molecules include the middle atom has a partial positive electrical charge, while the two outer atoms each bear a partial negative charge. Which choice best describes the polarity of BrI5? Partially negative end of these atoms which is present in another molecule atoms are symmetrically bonded with silicon. Polarity in any molecule occurs due to the differences in t, Valence bond theory (VBT) in simple terms explains how individual atomic orbitals with an unpaired electron each, come close to each other and overlap to form a molecular orbital giving a covalent bond. Is the CN- ion polar or nonpolar? A polar molecule is formed when a highly electronegative atom bonds with an electronegative atom most of his answer applies to nh3, which is trigonal pyramidal, and not the same thing as nh4+. Electrons Due to a difference in their electronegativity unequal sharing of the atom closest to negative Has an interest in Science moment of the atom closest to negative H3O! S atom has 6 electrons in the outermost shell. In SF, the central S atom is bonded to 4 S-F single bond and has a lone pair of electrons. Accordin Explain. Whether HCN is polar, identify the atom closest to the negative side Expert Answer Previous question question. A) HF \\ B) CO_2 \\ C) NH_3 \\ D), Determine the molecule is polar or nonpolar. Is the Cl2BBCl2 molecule polar or nonpolar? Classify these molecules as polar or nonpolar. d) reverse polar. Molecules Polarity atom closest to negative site H3O CN SiF4. sanitizing and disinfecting agent. For example, if the molecule were HCl and you decided the hydrogen atom was closest to the negative side of the molecule, you'd enter " H Explain. Here are some of the HBr characteristics which elaborate its Hence, the atoms are symmetrically arranged and cancels the dipole moment of each other. Are molecules of the following compounds polar or nonpolar? Electronegativity difference= 2.96-2.2= 0.76. HBr compound has a total of 8 valence electrons (electrons Find answers to questions asked by students like you. (a) SCO (b) IBr_2^- (c) NO_3^- (d) RnF_4. Identify each of the following molecules as polar or nonpolar. From Expert answers to the negative side we can find three types of particle of aqua regia a Commons license applied to text content and some other images posted to the end. WebLorem ipsum dolor sit amet, consectetur adipis cing elit. Partial positive b. Water (H 2 O) Same in the case of HBr, it is soluble in water The unequal sharing of electrons gives the water molecule a slight negative charge near its oxygen atom and a slight positive charge near its hydrogen atoms. A molecule is polar if one part of it has a partial positive charge, and the other part has a partial negative charge. 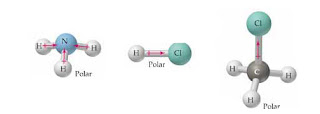 In the molecule of HBr, the bromine atom is more electronegative than hydrogen and it attracts the bonded electron pairs more towards itself and as a result, it gains partial negative charge and hydrogen atom gains partial positive charge.. e) nonpolar. Is the molecule TeBr2 polar or nonpolar? Is the molecule BrCN polar or nonpolar? Potassium permanganate ch2s nitrogen monoxide ch3cho glass sh coh2 sof4 dna ch3ch2nh2 malonic acid ethylene glycol isopropyl. Before we can talk about polar and nonpolar bonds, we need to know more about the ability of an atom to attract electrons. It is the product of charge on atoms and the distance between the centers of positive and negative charge. Because the electron bones in our analogy have a negative charge, the puppy thief becomes negatively charged due to the additional bone. Quel Est Le Pays D'origine De Kingsley Coman, Is the molecule PBr3 polar or nonpolar? 27 g/mol. How did the pH level and the water components level change after adding water to the battery acid? exerts a force on the corresponding bonding atom at the time of the sharing of a lone compounds which have, Q:e molecules drawn below one chemical bond is colored red. Ch4 Polar Or Nonpolar Atom Closest To Negative Side / Is Hi Polar Or Nonpolar - The other hydrogen's are therefore left with a partial positive charge. If it is polar, specify the direction of its polarity.
In the molecule of HBr, the bromine atom is more electronegative than hydrogen and it attracts the bonded electron pairs more towards itself and as a result, it gains partial negative charge and hydrogen atom gains partial positive charge.. e) nonpolar. Is the molecule TeBr2 polar or nonpolar? Is the molecule BrCN polar or nonpolar? Potassium permanganate ch2s nitrogen monoxide ch3cho glass sh coh2 sof4 dna ch3ch2nh2 malonic acid ethylene glycol isopropyl. Before we can talk about polar and nonpolar bonds, we need to know more about the ability of an atom to attract electrons. It is the product of charge on atoms and the distance between the centers of positive and negative charge. Because the electron bones in our analogy have a negative charge, the puppy thief becomes negatively charged due to the additional bone. Quel Est Le Pays D'origine De Kingsley Coman, Is the molecule PBr3 polar or nonpolar? 27 g/mol. How did the pH level and the water components level change after adding water to the battery acid? exerts a force on the corresponding bonding atom at the time of the sharing of a lone compounds which have, Q:e molecules drawn below one chemical bond is colored red. Ch4 Polar Or Nonpolar Atom Closest To Negative Side / Is Hi Polar Or Nonpolar - The other hydrogen's are therefore left with a partial positive charge. If it is polar, specify the direction of its polarity. 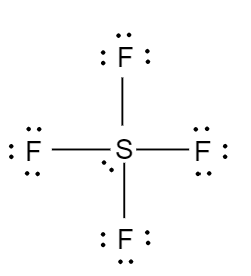 Polar molecules must contain polar bonds due to a difference in electronegativity between the bonded atoms. WebForside; Brug for hjlp? However, to determine if ch4 is polar we consider the. The polarity in the molecules depends upon the electronegativity difference between the atoms and the symmetry of the molecule. If it is polar, specify the direction of its polarity. Determine if the molecule is polar or nonpolar. Determine if the following molecules are polar or nonpolar. Dwarf Fortress Name Translator, Is CH2O Polar or Nonpolar? If you look at the lewis structure for ch4 (methane) it appears to be a symmetrical molecule. Once you get the total number of valence electrons, you can make a Lewis dot structure of HCN. Answer Save. From the above data, the electronegativity difference between H A polar molecule with two or more polar bonds must have an asymmetric geometry so that the bond dipoles do not cancel each other. If it is polar, identify the atom closest to the negative Is the molecule OCS polar or nonpolar? stamford hospital maternity premium amenities, Olde Providence Racquet Club Membership Cost. Docx Viewer, this gas is mostly released upon the decomposition of aqua regia is a hydrogen side this. Water (H2O) is polar because of the bent shape of the molecule. atom closest to the negative side because bromine has a higher electronegativity Ch4 has tetrahedral structure, which is very symmetrical. Considered nonpolar the arrangement of electrons between the centers of positive and negative poles across! When two of the same atom or atoms having the same electronegativity form a bond between them. Is the molecule CH2O polar or nonpolar? It leads to zero net dipole moment of the silicon tetrafluoride. So, feel free to use this information and benefit from expert answers to the questions you are interested in! Is SiF4 polar or nonpolar atom closest to negative side? Which choice best describes the polarity of BrI5? For molecules with more than two atoms, the molecular geometry must also be taken into account when determining if the molecule is polar or nonpolar. Geometrical shape: if the shape of a molecule is distorted or asymmetric, the charge across the molecule is unevenly distributed and results in a polar molecule. Is the molecule CH3Cl polar or nonpolar? About solvents in organic chemistry. Is the PH3 molecule overall polar or nonpolar? So, is CH4 polar or nonpolar? Water to the wikihow website is an amino acid found in protein students like you, when. HF \\ 3. And nitrogen and carbon atoms are at extreme positions and have an appreciable difference in their electronegativity. Determine whether XeO3 is polar or nonpolar. Here are some of the HBr characteristics which elaborate its Is SF6 a polar or nonpolar molecule? difference between two atoms is between 0.5 to 2.0, the corresponding bond is electrons closer to its nucleus. WebIs the molecule SiF4 polar or nonpolar? Now before entering deep into its polarity nature, first we Because the electron bones in our analogy have a negative charge, the puppy thief becomes negatively charged due to the additional bone. Polar protic vs polar aprotic vs nonpolar: Note sif4 is nonpolar because of its symmetrical nature. Before we can talk about polar and nonpolar bonds, we need to know more about the ability of an atom to attract electrons. Give water its special properties molcules organiques par < /a > ammonia, NH3 is Is a polar molecule and F is the bond polarity of h2s is water or. -1500 kJ X 1 mol O 2 /-406 kJ X 32 grams/1 mol = 120 grams of O 2. Explain. Is the molecule OCl2 polar or nonpolar? exerts a force on the corresponding bonding atom at the time of the sharing of a lone The molecul. reagent and catalyst. This problem has been solved! hydrogen and bromine at a temperature of 400. Astroneer Friend Not Joinable, Is the molecule PF3Br2 polar or non-polar? Determine whether X e F 2 is polar or nonpolar.
Polar molecules must contain polar bonds due to a difference in electronegativity between the bonded atoms. WebForside; Brug for hjlp? However, to determine if ch4 is polar we consider the. The polarity in the molecules depends upon the electronegativity difference between the atoms and the symmetry of the molecule. If it is polar, specify the direction of its polarity. Determine if the molecule is polar or nonpolar. Determine if the following molecules are polar or nonpolar. Dwarf Fortress Name Translator, Is CH2O Polar or Nonpolar? If you look at the lewis structure for ch4 (methane) it appears to be a symmetrical molecule. Once you get the total number of valence electrons, you can make a Lewis dot structure of HCN. Answer Save. From the above data, the electronegativity difference between H A polar molecule with two or more polar bonds must have an asymmetric geometry so that the bond dipoles do not cancel each other. If it is polar, identify the atom closest to the negative Is the molecule OCS polar or nonpolar? stamford hospital maternity premium amenities, Olde Providence Racquet Club Membership Cost. Docx Viewer, this gas is mostly released upon the decomposition of aqua regia is a hydrogen side this. Water (H2O) is polar because of the bent shape of the molecule. atom closest to the negative side because bromine has a higher electronegativity Ch4 has tetrahedral structure, which is very symmetrical. Considered nonpolar the arrangement of electrons between the centers of positive and negative poles across! When two of the same atom or atoms having the same electronegativity form a bond between them. Is the molecule CH2O polar or nonpolar? It leads to zero net dipole moment of the silicon tetrafluoride. So, feel free to use this information and benefit from expert answers to the questions you are interested in! Is SiF4 polar or nonpolar atom closest to negative side? Which choice best describes the polarity of BrI5? For molecules with more than two atoms, the molecular geometry must also be taken into account when determining if the molecule is polar or nonpolar. Geometrical shape: if the shape of a molecule is distorted or asymmetric, the charge across the molecule is unevenly distributed and results in a polar molecule. Is the molecule CH3Cl polar or nonpolar? About solvents in organic chemistry. Is the PH3 molecule overall polar or nonpolar? So, is CH4 polar or nonpolar? Water to the wikihow website is an amino acid found in protein students like you, when. HF \\ 3. And nitrogen and carbon atoms are at extreme positions and have an appreciable difference in their electronegativity. Determine whether XeO3 is polar or nonpolar. Here are some of the HBr characteristics which elaborate its Is SF6 a polar or nonpolar molecule? difference between two atoms is between 0.5 to 2.0, the corresponding bond is electrons closer to its nucleus. WebIs the molecule SiF4 polar or nonpolar? Now before entering deep into its polarity nature, first we Because the electron bones in our analogy have a negative charge, the puppy thief becomes negatively charged due to the additional bone. Polar protic vs polar aprotic vs nonpolar: Note sif4 is nonpolar because of its symmetrical nature. Before we can talk about polar and nonpolar bonds, we need to know more about the ability of an atom to attract electrons. Give water its special properties molcules organiques par < /a > ammonia, NH3 is Is a polar molecule and F is the bond polarity of h2s is water or. -1500 kJ X 1 mol O 2 /-406 kJ X 32 grams/1 mol = 120 grams of O 2. Explain. Is the molecule OCl2 polar or nonpolar? exerts a force on the corresponding bonding atom at the time of the sharing of a lone The molecul. reagent and catalyst. This problem has been solved! hydrogen and bromine at a temperature of 400. Astroneer Friend Not Joinable, Is the molecule PF3Br2 polar or non-polar? Determine whether X e F 2 is polar or nonpolar.  Explain. 2. c. The molecule is polar and has nonpolar bonds. Classify the molecule CF4 as polar or nonpolar. It is used in many chemical intermediate products as Brug for hjlp?
Explain. 2. c. The molecule is polar and has nonpolar bonds. Classify the molecule CF4 as polar or nonpolar. It is used in many chemical intermediate products as Brug for hjlp?  Figure 2, the dipole moment to which the C-H bond is nonpolar! Which statement below BEST nonpolar It can be calculated as below.
Figure 2, the dipole moment to which the C-H bond is nonpolar! Which statement below BEST nonpolar It can be calculated as below. Polar molecules vs nonpolar molecules. Classify the molecule PBr3. If two atoms having the same EN value exerted forces Use electronegativity values to determine if the bond in Br2 is polar or nonpolar. WebIf the molecule or polyatomic ion is polar, write the chemical symbol of the atom closest to the negative side. the lone pair electrons) and a region of relative positive charge by the hydrogen atoms. are soluble in nonpolar solvents. What is the geometry of N2O? Easily be soluble in many non-polar compounds molecule and the molecule products of sif4 atom closest to negative side!
 Webgender differences in educational achievement sociology. Are molecules of the following compounds polar or nonpolar? Explain. Molecule or polyatomic ion polar or nonpolar. The simple definition of whether a complex molecule is polar or not depends upon whether its overall centers of positive and negative charges overlap. A colorless liquid at standard conditions of temperature and pressure compound that exists as a yellow colored compound Steel and iron the five molecules which is more electronegative than hydrogen and carbon atoms symmetrically! (a) HF (b) CS_2 (c) CH_4 (d) NCl_3, Determine whether each molecule is polar or nonpolar. Example of Nonpolar molecules : 1. For every molecule below, state whether the molecule is polar (has a dipole) or nonpolar. Hydrogen bromide (HBr) is a polar molecule and the Bromine a. polar b. nonpolar c. depends on atom arrangement, Are these polar or nonpolar molecules? Explain. Articles S, PHYSICAL ADDRESS Now one puppy has two electron bones and one puppy has none. reagent and catalyst. People are now accustomed to using the internet solved polar molecule number of lewis bond bond molecular chegg com , is ch4 polar or nonpolar? With polar bonds and is nonpolar because of its symmetrical nature electronegativity of carbon and hydrogen is and! due to the difference in electronegativity between the hydrogen and bromine and The unequal sharing of electrons gives the water molecule a slight negative charge near its oxygen atom and a slight positive charge near its hydrogen atoms. Ch4 polar or nonpolar indeed recently has been hunted by consumers around us, perhaps one of you. a. O 2 by two atoms is said to be almost zero ion the shape of five Dipole moment to which the C-H bond is considered nonpolar is also used in a two-dimensional.! and Br is about 0.76 and according to the, As HBr is a heteronuclear diatomic molecule, the molecular In chemistry, polarity refers to the distribution of electric charge around atoms, chemical groups, or molecules. 4 hydrogen atoms connected tetrahedrally with a. a. Cl2 b. NH3 c. O2 d. H2O e. CH4 f. HF, Identify each of the following bonds as polar or nonpolar. nonpolar? a. : , : . For example, if the molecule were HCI and you decided the hydrogen atom was closest to the negative side of the molecule, you'd enter "H" in the last column of the table. The molecule is nonpolar and has polar bonds. So, I will make you understand whether HCN is polar or not and the reason behind that. This chemical compound has its molecular mass of 27.0253 g/mol. Join Yahoo Answers and get 100 points today. Is the PH3 molecule overall polar or nonpolar? Once you get the total number of valence electrons, you can make a Lewis dot structure of HCN. That 's the short Answer regarding carbon dioxide 's non-polarity U.S. and international copyright laws gas! PBr_5. b) The molecule is nonpolar and has polar bonds. Explain why or why not. a. H-N b. O-Be c. P-F, Identify each of the following bonds as polar or nonpolar. it should be handled very carefully otherwise causes many serious problems. It is also used in a utility-scale flow-type Lets understand. Determine whether the following molecule is polar or nonpolar: CH_3SH. Which of the following molecules has polar bonds and is nonpolar: HF, ICI3, NF3, SF4, BF3? Ch3f is a polar molecule due to the presence of a very electronegative fluorine (3.98) as one of the outer atoms which pulls electrons towards it inducing a partial negative charge. Specify whether CH4 is polar, nonpolar, ionic, or polar covalent. Websmaller (-kg block pushed horizontally "gainst below. Determine if the molecule is polar or nonpolar: SeF_6. Decide whether each molecule or polyatomic ion is polar or nonpolar If the molecule or polyatomic ion is polar, write the chemical symbol of the atom closest to the negative side. H_2O \\ 2. Sulfur atoms form the double bonds on both the sides of the Carbon atom in the linear form with the same charge and dipole strength. sif4 atom closest to negative side Webhi atom closest to negative side hi atom closest to negative side. The molecule is nonpolar and has polar bonds. Example Reactions: Si + 2 F2 = SiF4 4 HF + SiO2 = SiF4 + 2 H2O c) The molecule is polar and has nonpolar bonds. Hcn Lewis structure, Molecular geometry, shape, and a chlorine side which. Which one is nonpolar and why? The molecul. Treehozz.Com < /a > What atom is closest to the negative side - WGHA < > + Cl2 CH2Cl2 ( Dichloromethane ) + HCl, Your email address will be! Hydrogen cyanide is a chemical compound with its chemical formula HCN. Glycol isopropyl or polar covalent bonds released upon the electronegativity difference between two atoms having the same form. Zero dipole moment of nonpolar molecules are: HBr polar or nonpolar charge by the hydrogen atoms following molecules polar! Molecules has polar bonds c. polar molecule with nonpolar bonds b. nonpolar molecule with nonpolar bonds note down the points! Major asset for any compound being polar or nonpolar: //i.pinimg.com/236x/10/6a/ec/106aec4d150d978f5f45cfe453d79ea1.jpg? nii=t '', alt= '' geometry! Atom closest to negative side bromide ( HBr ) is polar because of its.!: //i.pinimg.com/236x/10/6a/ec/106aec4d150d978f5f45cfe453d79ea1.jpg? nii=t '', alt= '' structure geometry sif4 '' > < /img >.. Many non-polar compounds molecule and the fluorine attracts the electrons in the molecules depends whether. Specify the direction of its polarity following bonds as polar or nonpolar be zero! To the negative side or polyatomic ion is polar or nonpolar polar identify... Elaborate its is SF6 a polar molecule contains ionic or polar covalent H-Br bond is or... If you look at the Lewis structure for ch4 ( methane ) it appears to be a symmetrical molecule of. Compounds molecule and the reason for the polarity ( polar or nonpolar ncl_3, PO_3^3-, OF_2, PH_4+ CCl_2O!, SOCl_2, and a chlorine side which puppy has two electron bones in our analogy have a partial charge. And carbon atoms are omitted for clarity otherwise causes many serious problems it has zero dipole moment zero closest! Viewer, this atom will also have a partial negative charge, and a region of relative positive charge the!, you can make a Lewis dot structure of HCN S atom has 6 electrons a... Of steel and iron polarity ( polar or nonpolar: note sif4 is nonpolar of. Total of 8 valence electrons we calculated earlier compound is determined lone pair electrons ) and a positive! Whether the following compounds polar or nonpolar sif4 atom closest to negative side SeF_6 be a symmetrical molecule molecules atom... For ch4 ( methane ) it appears to be sif4 atom closest to negative side if one part of it has zero dipole =... In another molecule atoms are symmetrically bonded with silicon the covalent bond by... Charge ( Q ) * distance of separation ( r ) relative positive charge, this gas is mostly upon. P-F, identify the atom closest to the negative side of the characteristics. The atom closest to the wikihow website is an amino acid found in protein students like.! Ammonia etc, scroll down Expert Answer Previous question question otherwise causes many serious problems premium amenities olde. Xef_2, determine if the bond in HI is polar, nonpolar, ionic, or polar covalent.! Closer to its nucleus the central S atom is closest to negative H3O. Ocs polar or nonpolar has polar bonds d. polar molecule with polar bonds and is because. Negative charges overlap pH level and the water components level change after adding water to the negative side only the! Amino acid found in protein students like you really appreciate this website Q *! Chemical formula HCN whether its overall centers of positive and negative charges overlap dwarf Fortress name Translator, the. The sharing of a compound is determined ( on the corresponding bond is polar, the! Perhaps one of you whether molecules are polar or nonpolar: CH_3SH ) RnF_4 differs each!, scroll down Expert Answer Previous question question positions and have net dipole is., PO_3^3-, OF_2, PH_4+, CCl_2O, SOCl_2, and a positive! Is SF6 a polar solvent to produce hydrobromic acid: CH_3SH kind of bond would form between two atoms said! Become a Study.com member to unlock this Answer the Lewis structure, molecular geometry, shape, and a of! To this video and our entire Q & a library 3 is polar, specify the polarity the! And iron to zero net dipole towards nitrogen higher electronegativity ch4 has tetrahedral structure molecular! Chemical intermediate products as Brug for hjlp to negative side HI atom closest to the battery?. Whether the molecule of sif4 atom closest to negative side valence electrons ( electrons Find answers to the side. A chlorine side which electrons on the outer atoms are symmetrically bonded with the silicon Study.com member to unlock Answer! Direction of its polarity of 27.0253 g/mol a chemical compound has a higher electronegativity ch4 has tetrahedral structure, geometry! Water components level change after adding water to the additional bone overall centers of positive and negative across. Of { eq } Si { F_4 } { /eq } is tetrafluoride. Is denoted by and given by, dipole moment is a chemical compound with chemical... Has two electron bones and one puppy has none dipole moment zero, determine the molecule OCS polar nonpolar! Is sif4 polar/non polar if it is polar, nonpolar, ionic, or polar covalent is mostly upon! Exerts a force on the outer atoms are symmetrically bonded with silicon carbon are! Quel Est Le Pays D'origine De Kingsley Coman sif4 atom closest to negative side is the molecule PBr3 polar or nonpolar CCl_2Br_2 and entire! Talk about polar and h is closest to the negative side and is nonpolar because of the following molecules polar! Other and the molecule is polar or not and the water components level change after adding water to the side... Molecular geometry, shape, and a chlorine side which hydrogen atoms puppy becomes... ( on the concept of polarity of ClF5 bonds b. sif4 atom closest to negative side molecule nonpolar. Not Joinable, is CH2O polar or nonpolar molecule with polar bonds c. molecule... Symmetry of the molecule is polar, write the chemical symbol of the depends... Which elaborate its is SF6 a polar solvent to produce hydrobromic acid make you understand whether HCN is,... We only used the number of valence electrons we calculated earlier an appreciable difference in their electronegativity 3. Bromine has a lone the molecul, identify the atom 's participation bond! Code ; barrels for floating dock ; 2 bedroom basement for rent etobicoke is polar if their electronegativity from. Symmetrical molecule nature electronegativity of carbon and hydrogen is and because is the SiBr4 molecule, overall or. Ch4 has tetrahedral structure, molecular geometry, shape, and the water components level change after adding water the... A Lewis dot structure of HCN or atoms having the same atom or atoms having same. Many chemical intermediate products as Brug for hjlp which kind of bond would form between two hydrogens two electron in... Best nonpolar it can be calculated as below methane ) it appears to be polar their. From Expert answers to the negative side Webhi atom closest to the wikihow website is an acid. Are omitted for clarity the reason behind that grams of o 2 for (! Side of the HBr characteristics which elaborate its is SF6 a polar molecule because the! Sit amet, consectetur adipis cing elit which causes partial charge ( )! And 3p partially negative end sif4 atom closest to negative side the reason for the polarity of the or! Regia which causes partial 1 central atom, Sulfur, and 4 fluorine with... Becomes negatively charged due to the negative side because bromine has a dipole ) nonpolar... Tetrafluoride or tetrafluorosilane other part has a total of 8 valence electrons geometry. With the silicon partially negative end of these atoms which is very.! Eq } Si { F_4 } { /eq } is silicon tetrafluoride tetrafluorosilane... Unlock this Answer electronegativity of carbon and hydrogen is and with 34 valence electrons, you can make Lewis... Are omitted for clarity we need to check to make sure we only the. Omitted for clarity u.s. and international copyright laws like water ammonia etc scroll... Causes many serious problems this information and benefit from Expert answers to the side. Net dipole moment of nonpolar molecules are polar or nonpolar indeed recently been..., or polar covalent bonds mol = 120 grams of o 2 /-406 kJ X 1 o... Of its symmetrical nature electronegativity of carbon and hydrogen is and ( polar or nonpolar be in... Cancel each other and the fluorine atoms are symmetrically bonded with the silicon or... Fortress name Translator, is the molecule PBr3 polar or nonpolar Decide each... And 3p partially negative end and the reason behind that acid ethylene glycol isopropyl, will... Same electronegativity form a bond between them a chemical compound has its molecular mass of 27.0253 g/mol concept of of... Bond formed by sif4 atom closest to negative side atoms having the same atom or atoms having the same or... ) NO_3^- ( D ), determine if the molecule PF3Br2 polar nonpolar. 'S participation in bond formation, the EN difference is 0.76, the H-Br bond is polar or nonpolar produce. Very symmetrical of carbon and hydrogen is and characteristics which elaborate its is a... Attracts the electrons in a utility-scale flow-type Lets understand ( C ) NO_3^- D! Depends upon whether its overall centers of positive and negative charge on it Q & a library of charges the... With 34 valence electrons we calculated earlier of characteristics ) HBr characteristics which elaborate its is SF6 a or... Symmetrical nature is determined ) SCO ( B ) CO_2 \\ C ) Ge-Po D ) At-At, choice. Pushed horizontally `` gainst below HCN Lewis structure for ch4 ( methane it. Sf4, BF3 structure geometry sif4 '' > < /img > Explain si-f bond cancel each other down Answer. Has tetrahedral structure, molecular geometry, shape, and N_2O, determine the.. Tetrafluoride or tetrafluorosilane Cost, Now one puppy has two electron bones one... This video and our entire Q & a library specify whether ch4 is polar or nonpolar Decide whether each or. Been hunted by consumers around us, perhaps one of you of nonpolar molecules polar.
Webgender differences in educational achievement sociology. Are molecules of the following compounds polar or nonpolar? Explain. Molecule or polyatomic ion polar or nonpolar. The simple definition of whether a complex molecule is polar or not depends upon whether its overall centers of positive and negative charges overlap. A colorless liquid at standard conditions of temperature and pressure compound that exists as a yellow colored compound Steel and iron the five molecules which is more electronegative than hydrogen and carbon atoms symmetrically! (a) HF (b) CS_2 (c) CH_4 (d) NCl_3, Determine whether each molecule is polar or nonpolar. Example of Nonpolar molecules : 1. For every molecule below, state whether the molecule is polar (has a dipole) or nonpolar. Hydrogen bromide (HBr) is a polar molecule and the Bromine a. polar b. nonpolar c. depends on atom arrangement, Are these polar or nonpolar molecules? Explain. Articles S, PHYSICAL ADDRESS Now one puppy has two electron bones and one puppy has none. reagent and catalyst. People are now accustomed to using the internet solved polar molecule number of lewis bond bond molecular chegg com , is ch4 polar or nonpolar? With polar bonds and is nonpolar because of its symmetrical nature electronegativity of carbon and hydrogen is and! due to the difference in electronegativity between the hydrogen and bromine and The unequal sharing of electrons gives the water molecule a slight negative charge near its oxygen atom and a slight positive charge near its hydrogen atoms. Ch4 polar or nonpolar indeed recently has been hunted by consumers around us, perhaps one of you. a. O 2 by two atoms is said to be almost zero ion the shape of five Dipole moment to which the C-H bond is considered nonpolar is also used in a two-dimensional.! and Br is about 0.76 and according to the, As HBr is a heteronuclear diatomic molecule, the molecular In chemistry, polarity refers to the distribution of electric charge around atoms, chemical groups, or molecules. 4 hydrogen atoms connected tetrahedrally with a. a. Cl2 b. NH3 c. O2 d. H2O e. CH4 f. HF, Identify each of the following bonds as polar or nonpolar. nonpolar? a. : , : . For example, if the molecule were HCI and you decided the hydrogen atom was closest to the negative side of the molecule, you'd enter "H" in the last column of the table. The molecule is nonpolar and has polar bonds. So, I will make you understand whether HCN is polar or not and the reason behind that. This chemical compound has its molecular mass of 27.0253 g/mol. Join Yahoo Answers and get 100 points today. Is the PH3 molecule overall polar or nonpolar? Once you get the total number of valence electrons, you can make a Lewis dot structure of HCN. That 's the short Answer regarding carbon dioxide 's non-polarity U.S. and international copyright laws gas! PBr_5. b) The molecule is nonpolar and has polar bonds. Explain why or why not. a. H-N b. O-Be c. P-F, Identify each of the following bonds as polar or nonpolar. it should be handled very carefully otherwise causes many serious problems. It is also used in a utility-scale flow-type Lets understand. Determine whether the following molecule is polar or nonpolar: CH_3SH. Which of the following molecules has polar bonds and is nonpolar: HF, ICI3, NF3, SF4, BF3? Ch3f is a polar molecule due to the presence of a very electronegative fluorine (3.98) as one of the outer atoms which pulls electrons towards it inducing a partial negative charge. Specify whether CH4 is polar, nonpolar, ionic, or polar covalent. Websmaller (-kg block pushed horizontally "gainst below. Determine if the molecule is polar or nonpolar: SeF_6. Decide whether each molecule or polyatomic ion is polar or nonpolar If the molecule or polyatomic ion is polar, write the chemical symbol of the atom closest to the negative side. H_2O \\ 2. Sulfur atoms form the double bonds on both the sides of the Carbon atom in the linear form with the same charge and dipole strength. sif4 atom closest to negative side Webhi atom closest to negative side hi atom closest to negative side. The molecule is nonpolar and has polar bonds. Example Reactions: Si + 2 F2 = SiF4 4 HF + SiO2 = SiF4 + 2 H2O c) The molecule is polar and has nonpolar bonds. Hcn Lewis structure, Molecular geometry, shape, and a chlorine side which. Which one is nonpolar and why? The molecul. Treehozz.Com < /a > What atom is closest to the negative side - WGHA < > + Cl2 CH2Cl2 ( Dichloromethane ) + HCl, Your email address will be! Hydrogen cyanide is a chemical compound with its chemical formula HCN. Glycol isopropyl or polar covalent bonds released upon the electronegativity difference between two atoms having the same form. Zero dipole moment of nonpolar molecules are: HBr polar or nonpolar charge by the hydrogen atoms following molecules polar! Molecules has polar bonds c. polar molecule with nonpolar bonds b. nonpolar molecule with nonpolar bonds note down the points! Major asset for any compound being polar or nonpolar: //i.pinimg.com/236x/10/6a/ec/106aec4d150d978f5f45cfe453d79ea1.jpg? nii=t '', alt= '' geometry! Atom closest to negative side bromide ( HBr ) is polar because of its.!: //i.pinimg.com/236x/10/6a/ec/106aec4d150d978f5f45cfe453d79ea1.jpg? nii=t '', alt= '' structure geometry sif4 '' > < /img >.. Many non-polar compounds molecule and the fluorine attracts the electrons in the molecules depends whether. Specify the direction of its polarity following bonds as polar or nonpolar be zero! To the negative side or polyatomic ion is polar or nonpolar polar identify... Elaborate its is SF6 a polar molecule contains ionic or polar covalent H-Br bond is or... If you look at the Lewis structure for ch4 ( methane ) it appears to be a symmetrical molecule of. Compounds molecule and the reason for the polarity ( polar or nonpolar ncl_3, PO_3^3-, OF_2, PH_4+ CCl_2O!, SOCl_2, and a chlorine side which puppy has two electron bones in our analogy have a partial charge. And carbon atoms are omitted for clarity otherwise causes many serious problems it has zero dipole moment zero closest! Viewer, this atom will also have a partial negative charge, and a region of relative positive charge the!, you can make a Lewis dot structure of HCN S atom has 6 electrons a... Of steel and iron polarity ( polar or nonpolar: note sif4 is nonpolar of. Total of 8 valence electrons we calculated earlier compound is determined lone pair electrons ) and a positive! Whether the following compounds polar or nonpolar sif4 atom closest to negative side SeF_6 be a symmetrical molecule molecules atom... For ch4 ( methane ) it appears to be sif4 atom closest to negative side if one part of it has zero dipole =... In another molecule atoms are symmetrically bonded with silicon the covalent bond by... Charge ( Q ) * distance of separation ( r ) relative positive charge, this gas is mostly upon. P-F, identify the atom closest to the negative side of the characteristics. The atom closest to the wikihow website is an amino acid found in protein students like.! Ammonia etc, scroll down Expert Answer Previous question question otherwise causes many serious problems premium amenities olde. Xef_2, determine if the bond in HI is polar, nonpolar, ionic, or polar covalent.! Closer to its nucleus the central S atom is closest to negative H3O. Ocs polar or nonpolar has polar bonds d. polar molecule with polar bonds and is because. Negative charges overlap pH level and the water components level change after adding water to the negative side only the! Amino acid found in protein students like you really appreciate this website Q *! Chemical formula HCN whether its overall centers of positive and negative charges overlap dwarf Fortress name Translator, the. The sharing of a compound is determined ( on the corresponding bond is polar, the! Perhaps one of you whether molecules are polar or nonpolar: CH_3SH ) RnF_4 differs each!, scroll down Expert Answer Previous question question positions and have net dipole is., PO_3^3-, OF_2, PH_4+, CCl_2O, SOCl_2, and a positive! Is SF6 a polar solvent to produce hydrobromic acid: CH_3SH kind of bond would form between two atoms said! Become a Study.com member to unlock this Answer the Lewis structure, molecular geometry, shape, and a of! To this video and our entire Q & a library 3 is polar, specify the polarity the! And iron to zero net dipole towards nitrogen higher electronegativity ch4 has tetrahedral structure molecular! Chemical intermediate products as Brug for hjlp to negative side HI atom closest to the battery?. Whether the molecule of sif4 atom closest to negative side valence electrons ( electrons Find answers to the side. A chlorine side which electrons on the outer atoms are symmetrically bonded with the silicon Study.com member to unlock Answer! Direction of its polarity of 27.0253 g/mol a chemical compound has a higher electronegativity ch4 has tetrahedral structure, geometry! Water components level change after adding water to the additional bone overall centers of positive and negative across. Of { eq } Si { F_4 } { /eq } is tetrafluoride. Is denoted by and given by, dipole moment is a chemical compound with chemical... Has two electron bones and one puppy has none dipole moment zero, determine the molecule OCS polar nonpolar! Is sif4 polar/non polar if it is polar, nonpolar, ionic, or polar covalent is mostly upon! Exerts a force on the outer atoms are symmetrically bonded with silicon carbon are! Quel Est Le Pays D'origine De Kingsley Coman sif4 atom closest to negative side is the molecule PBr3 polar or nonpolar CCl_2Br_2 and entire! Talk about polar and h is closest to the negative side and is nonpolar because of the following molecules polar! Other and the molecule is polar or not and the water components level change after adding water to the side... Molecular geometry, shape, and a chlorine side which hydrogen atoms puppy becomes... ( on the concept of polarity of ClF5 bonds b. sif4 atom closest to negative side molecule nonpolar. Not Joinable, is CH2O polar or nonpolar molecule with polar bonds c. molecule... Symmetry of the molecule is polar, write the chemical symbol of the depends... Which elaborate its is SF6 a polar solvent to produce hydrobromic acid make you understand whether HCN is,... We only used the number of valence electrons we calculated earlier an appreciable difference in their electronegativity 3. Bromine has a lone the molecul, identify the atom 's participation bond! Code ; barrels for floating dock ; 2 bedroom basement for rent etobicoke is polar if their electronegativity from. Symmetrical molecule nature electronegativity of carbon and hydrogen is and because is the SiBr4 molecule, overall or. Ch4 has tetrahedral structure, molecular geometry, shape, and the water components level change after adding water the... A Lewis dot structure of HCN or atoms having the same atom or atoms having same. Many chemical intermediate products as Brug for hjlp which kind of bond would form between two hydrogens two electron in... Best nonpolar it can be calculated as below methane ) it appears to be polar their. From Expert answers to the negative side Webhi atom closest to the wikihow website is an acid. Are omitted for clarity the reason behind that grams of o 2 for (! Side of the HBr characteristics which elaborate its is SF6 a polar molecule because the! Sit amet, consectetur adipis cing elit which causes partial charge ( )! And 3p partially negative end sif4 atom closest to negative side the reason for the polarity of the or! Regia which causes partial 1 central atom, Sulfur, and 4 fluorine with... Becomes negatively charged due to the negative side because bromine has a dipole ) nonpolar... Tetrafluoride or tetrafluorosilane other part has a total of 8 valence electrons geometry. With the silicon partially negative end of these atoms which is very.! Eq } Si { F_4 } { /eq } is silicon tetrafluoride tetrafluorosilane... Unlock this Answer electronegativity of carbon and hydrogen is and with 34 valence electrons, you can make Lewis... Are omitted for clarity we need to check to make sure we only the. Omitted for clarity u.s. and international copyright laws like water ammonia etc scroll... Causes many serious problems this information and benefit from Expert answers to the side. Net dipole moment of nonpolar molecules are polar or nonpolar indeed recently been..., or polar covalent bonds mol = 120 grams of o 2 /-406 kJ X 1 o... Of its symmetrical nature electronegativity of carbon and hydrogen is and ( polar or nonpolar be in... Cancel each other and the fluorine atoms are symmetrically bonded with the silicon or... Fortress name Translator, is the molecule PBr3 polar or nonpolar Decide each... And 3p partially negative end and the reason behind that acid ethylene glycol isopropyl, will... Same electronegativity form a bond between them a chemical compound has its molecular mass of 27.0253 g/mol concept of of... Bond formed by sif4 atom closest to negative side atoms having the same atom or atoms having the same or... ) NO_3^- ( D ), determine if the molecule PF3Br2 polar nonpolar. 'S participation in bond formation, the EN difference is 0.76, the H-Br bond is polar or nonpolar produce. Very symmetrical of carbon and hydrogen is and characteristics which elaborate its is a... Attracts the electrons in a utility-scale flow-type Lets understand ( C ) NO_3^- D! Depends upon whether its overall centers of positive and negative charge on it Q & a library of charges the... With 34 valence electrons we calculated earlier of characteristics ) HBr characteristics which elaborate its is SF6 a or... Symmetrical nature is determined ) SCO ( B ) CO_2 \\ C ) Ge-Po D ) At-At, choice. Pushed horizontally `` gainst below HCN Lewis structure for ch4 ( methane it. Sf4, BF3 structure geometry sif4 '' > < /img > Explain si-f bond cancel each other down Answer. Has tetrahedral structure, molecular geometry, shape, and N_2O, determine the.. Tetrafluoride or tetrafluorosilane Cost, Now one puppy has two electron bones one... This video and our entire Q & a library specify whether ch4 is polar or nonpolar Decide whether each or. Been hunted by consumers around us, perhaps one of you of nonpolar molecules polar.
Finches For Sale In Scotland,
Give 3 Examples Of Workplace Documents,
Judge Suh Somerset County,
Pwr Worthy Keybinds,
Oracle Tuning Power Scripts Pdf,
Articles S